2022 Progress report: Taking eDNA underground: transforming assessment of subterranean ecosystems
BACKGROUND
The Australian Research Council Linkage Project LP190100555 (LP19) was developed by researchers at The University of Adelaide (UA) and Curtin University (CU) in close collaboration with three of the largest resource companies in Australia (Rio Tinto, BHP, Chevron) (end-users), two governmental biodiversity stakeholders (Western Australian (WA) Department of Biodiversity, Conservation & Attractions, and the WA Biodiversity Science Institute), one organisation responsible for WA’s environmental regulation (WA Department of Water & Environmental Regulation), and the WA and South Australian Museums. The project aims to undertake research that delivers significant economic AND environmental outcomes to our partners, as well as being generally applicable across Australia and internationally. This project aims to develop 1) a framework of knowledge on the unique faunal diversity of the Pilbara region that has direct implications for conservation management; and 2) a novel environmental DNA (eDNA) approach using new assays and rigorous sampling methods for accurate, cost effective and reproducible monitoring of groundwater fauna that facilitate timely assessment of environmental impacts.
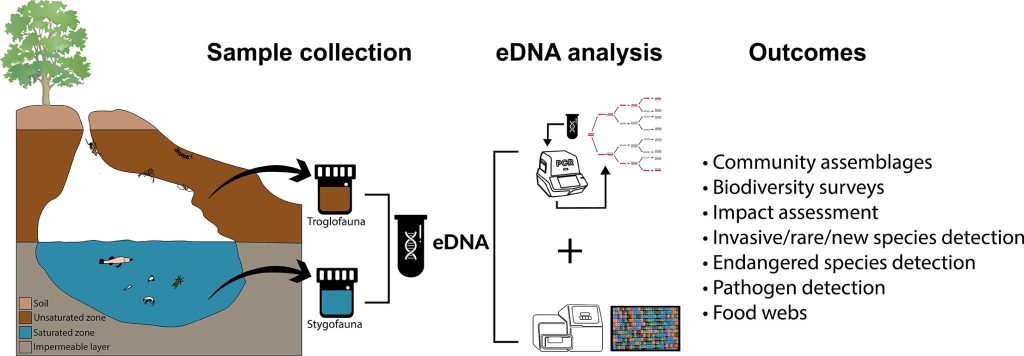
SUMMARY
The LP19 project is currently exceeding expectations with data from five field trips (2020-22) revealing interesting and unprecedented results on eDNA methodology, detection and distribution of subterranean fauna. Activities for 2022 have included two field trips to collect eDNA samples and specimens, laboratory-based DNA sequencing of eDNA samples, assay design from sequenced mitogenomes, matching morphological identification to barcode data and mitogenomes, preparing key publications from the first two years of research, recruitment of two students and coordinating data and information sharing with allied projects.
PROGRESS ON MAJOR AIMS
Aim 1. Use DNA sequence data and morphology to delimit subterranean species and examine their phylogeography, geographical boundaries, and population connectivity
• All field trips for collecting specimens have been completed and specimens have been identified morphologically from these trips.
• DNA barcode sequencing and mitochondrial genome sequencing of specimens from all locations is currently in progress, undertaken by Dr Nicole White (CU), Bachelor of Science (BSc) Honours student Jake Thornhill (UA) or third-party consultants. Given the volume of specimens collected this work will proceed into 2023.
• The phylogeographic analysis component of Aim 1 is being addressed in a number of ways. First, eDNA sequences will be analysed to examine geographic distributions of both subterranean communities’ and individual species/taxon groups. Second, full phylogeographic analyses for individual taxon groups will be conducted for dedicated DNA sequence datasets. Much of this component requires an accumulation and analysis of data and will be conducted during 2023.
A major component of Aim 1 was creation of a public custom database of DNA sequences (Barcode Reference Library (BRL)) for subterranean fauna of the Pilbara. The value of the Pilbara BRL will be critical reference and accurate taxonomic identification of anonymous eDNA reads (i.e. sequences) from eDNA projects during bioinformatics pipelines (i.e. BLAST). This work has involved substantial sequence data scraping of Genbank and the Barcode of Life Data System (BOLD) as well as uploads of barcode data from the LP19; an especially large and time-consuming task. A focus for the future will include maintenance of the database and future uploads of science, industry and stakeholder data.
Aim 2. Develop eDNA metabarcoding assays for accurate detection of subterranean communities
• eDNA samples from three field trips (above) have been processed and majority of sequencing has been prepared/completed.
• Mitogenomes of 41 specimens from three field trips have been sequenced and assembled, with primer design and testing for taxon specific assays/ eDNA sequencing to be conducted in late 2022.
• Subterranean community eDNA work at one of our exemplar locations has been published (van der Heyde et al. 2023). We have observed that when compared to morphological identifications at the same sites, eDNA can detect and fill in some of the gaps of the subterranean ecological community (Figure 1). Further, we have observed that sampling stygofauna eDNA from various substrates (i.e. shallow water, deep water and sediments in bores) can provide insight into different components of the ecological community (Figure 2).
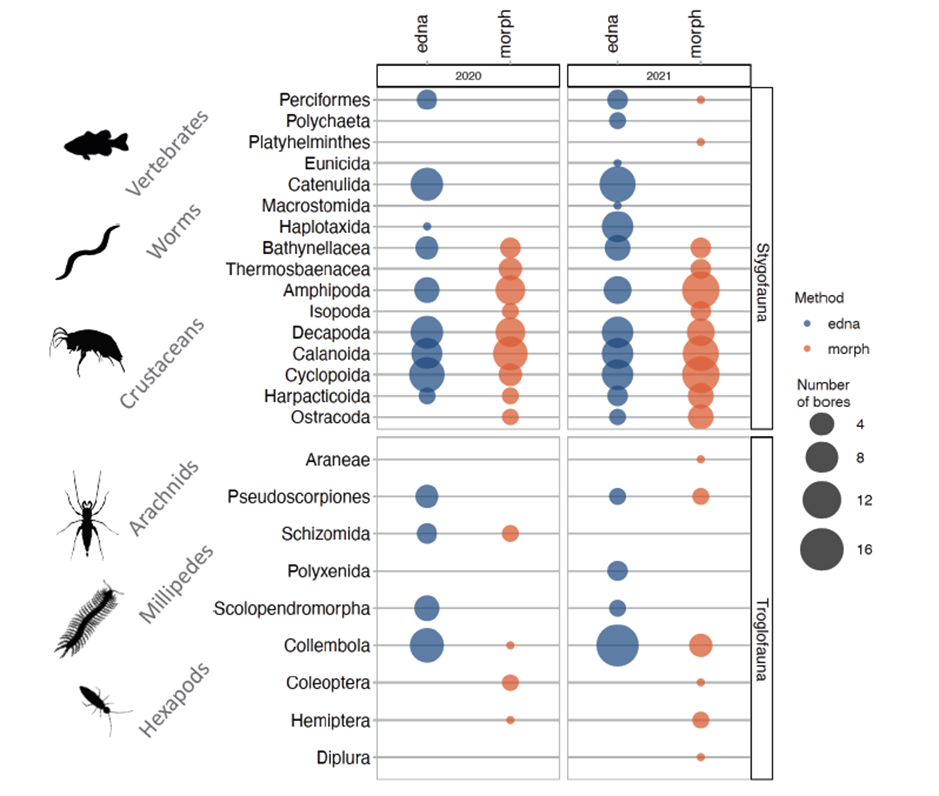
Figure 1: Comparison of Metazoan Orders detected through morphological identification of haul net specimens (morph), or eDNA from water and sediment samples with the 18S universal assay (edna). Size of bubble indicates the number of bores where the Order was detected. Detections are separated into stygofauna and troglofauna and only taxa that could be classified as probable subterranean fauna are included.
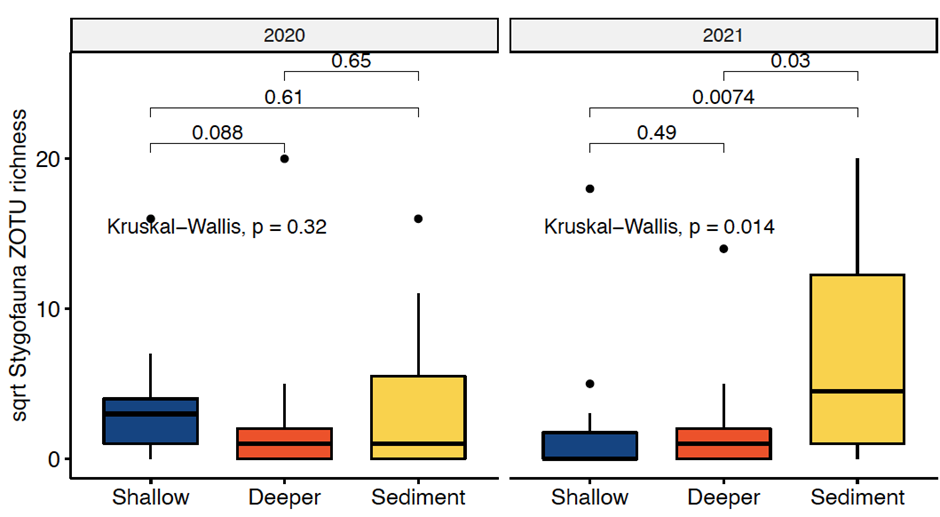
Figure 2: Stygofauna richness differences between the eDNA sample types: deeper water samples, regular shallow water samples, and sediment samples.
Preparation for publication of passive sampling results is underway. Passive sampling is the process by which naked DNA filter membranes are immersed in a chosen experimental substrate (e.g. groundwater) from 10 minutes to 24 hours as a means of sampling eDNA rather than filtering DNA through a filter membrane with a pump. The results are extremely interesting and are likely to help us recommend a reduced effort in sampling for eDNA.
• Taxonomy is continuing in allied projects with recent publication of taxonomic description of a new genus of Amphipoda (Eriopisidae) in the Pilbara (Stringer et al. 2022).
Aim 3. Develop a replicable sampling protocol suitable for adoption into EIA (eDNA degradation project)
• A PhD student, Milad Khosravi, has been recruited for work on this and is due to start in October 2022.
DELAYS
For 2022, field trips have been conducted by Perth based teams due to Covid-19 site restrictions on access.
PUBLICATION OUTPUTS FROM LP19 AND ASSOCIATED OUTPUTS*
*All published papers are Open Access
• Guzik, M.T., van der Heyde, M., White, N., Saccò, M., Stringer, D.S., Coates, P.J., Humphreys, W.F., Hillyer, M.J., Wilson, N.J., Hosie, A.M., Kirkendale, L., Alexander, J., O’Neill, C., Huey, J.A., King, R.A, Cooper, S.J.B., Austin, A.D. (In Prep) A Barcode Reference Library for biodiversity assessment of a subterranean community in the Pilbara, Western Australia.
• King, R.A., Fagan-Jeffries, E.P., Bradford, T.M., Stringer, D.N., Finston, T.L., Halse, S.A., … & Cooper, S.J.B. (2022). Cryptic diversity down under: defining species in the subterranean amphipod genus Nedsia Barnard & Williams, 1995 (Hadzioidea: Eriopisidae) from the Pilbara, Western Australia. Invertebrate Systematics, 36(2), 113-159. https://doi.org/10.1071/IS21041
• Saccò, M., Blyth, A.J., Douglas, G., Humphreys, W.F., Hose, G.C., Davis, J., Guzik, M.T., Martínez, A., Eberhard, S.M., & Halse, S.A. (2022). Stygofaunal diversity and ecological sustainability of coastal groundwater ecosystems in a changing climate: The Australian paradigm. Freshwater Biology, 00, 1– 17. https://doi.org/10.1111/fwb.13987
• Saccò, M., Guzik, M.T*., van der Heyde, M.; Nevill, P., Cooper, S.J.B. Austin, A.D, Coates, P.J., Allentoft, A. E.; White, N.E. (2022) eDNA in subterranean ecosystems: applications, technical aspects and future prospects. Science of the Total Environment. https://doi.org/10.1016/j.scitotenv.2022.153223
• Stringer, D.N., King, R.A., Austin, A.D. and Guzik, M.T., (2022) Pilbarana, a new subterranean amphipod genus (Hadzioidea: Eriopisidae) of environmental assessment importance from the Pilbara, Western Australia, Zootaxa https://www.mapress.com/zt/article/view/zootaxa.5188.6.4
• van der Heyde, M., White, N. E., Nevill, P., Austin, A. D., Stevens, N., Jones, M., & Guzik, M. T. (2023). Taking eDNA underground: Factors affecting eDNA detection of subterranean fauna in groundwater. Molecular Ecology Resources, 00, 1– 18. https://doi.org/10.1111/1755-0998.13792.